Ustekinumab is a fully human IgG1κ monoclonal antibody that has transformed the management of immune-mediated inflammatory diseases through selective inhibition of interleukin signaling. By targeting the shared p40 subunit of IL-12 and IL-23, it modulates Th1 and Th17 pathways central to chronic inflammation.
For medical educators, rheumatologists, gastroenterologists, and clinical researchers, communicating both molecular specificity and therapeutic outcomes requires educational tools that integrate immunology, pharmacology, and clinical trial evidence.
The Ustekinumab PowerPoint Presentation Template provides a structured visual framework to support this multidimensional teaching approach.
Below is a guide for leveraging the template across academic and clinical settings.
1. Molecular Foundations: Understanding Monoclonal Antibody Architecture Effective biologic education begins with molecular anatomy.
Antibody Structure Use this slide to identify:
Heavy and light chains Variable regions responsible for antigen binding Constant regions mediating immune effector functions Antibody Structure Slide from Ustekinumab PowerPoint Presentation Template This establishes the structural basis of antigen specificity.
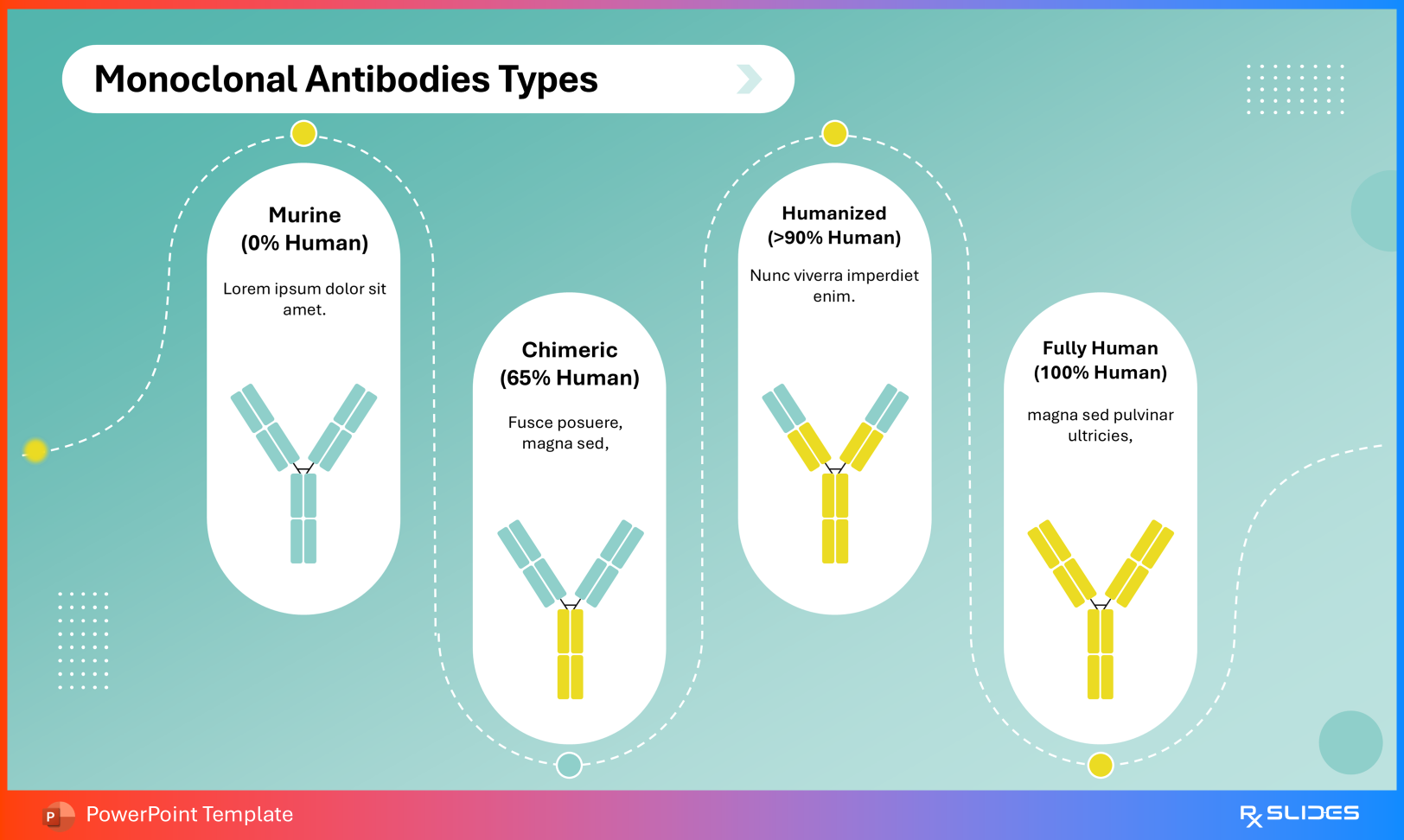
Monoclonal Antibody Classification The Monoclonal Antibodies Types slide visually differentiates:
Murine (0% human) Chimeric (~65% human) Humanized (>90% human) Fully human (100% human) Monoclonal Antibodies Types Slide from Ustekinumab PowerPoint Presentation Template This progression contextualizes immunogenicity risk and therapeutic evolution.
Ustekinumab Antibody Type Conclude with confirmation that ustekinumab is a fully human monoclonal antibody, supporting its favorable tolerability profile.
Ustekinumab antibody type Slide from Ustekinumab PowerPoint Presentation Template 2. Mechanism of Action: Targeting IL-12 and IL-23 Signaling For immunology-focused audiences, visualizing cytokine interactions is essential.
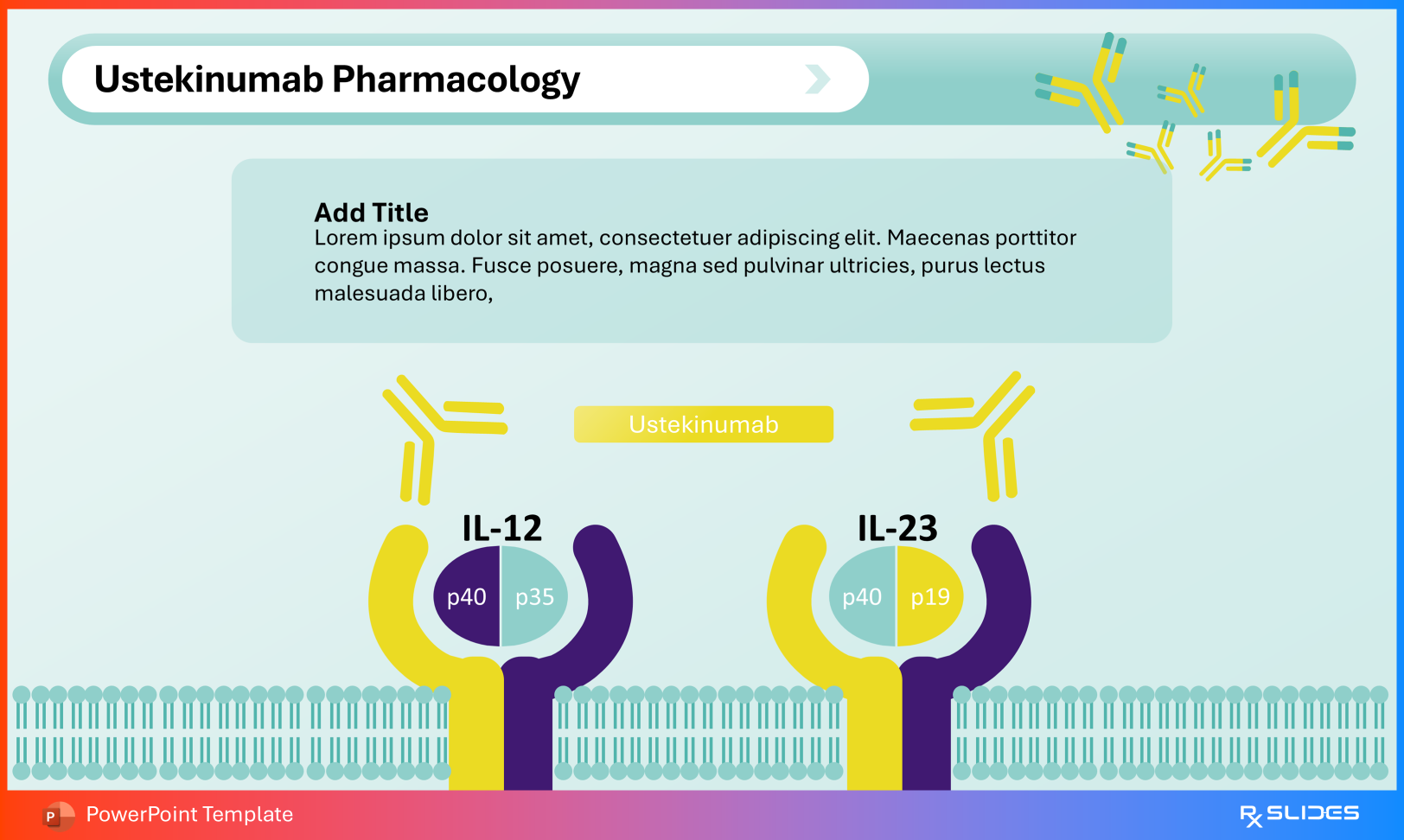
Ustekinumab Pharmacology Use this slide to illustrate:
IL-12 composed of p35 + p40 subunits IL-23 composed of p19 + p40 subunits The shared p40 subunit represents ustekinumab’s therapeutic target.
Ustekinumab Pharmacology Slide from Ustekinumab PowerPoint Presentation Template 3. Clinical Indications and Disease-Specific Mechanisms For clinicians and residents, application to disease pathology becomes central.
Ustekinumab Indications The template organizes FDA-approved uses, including:
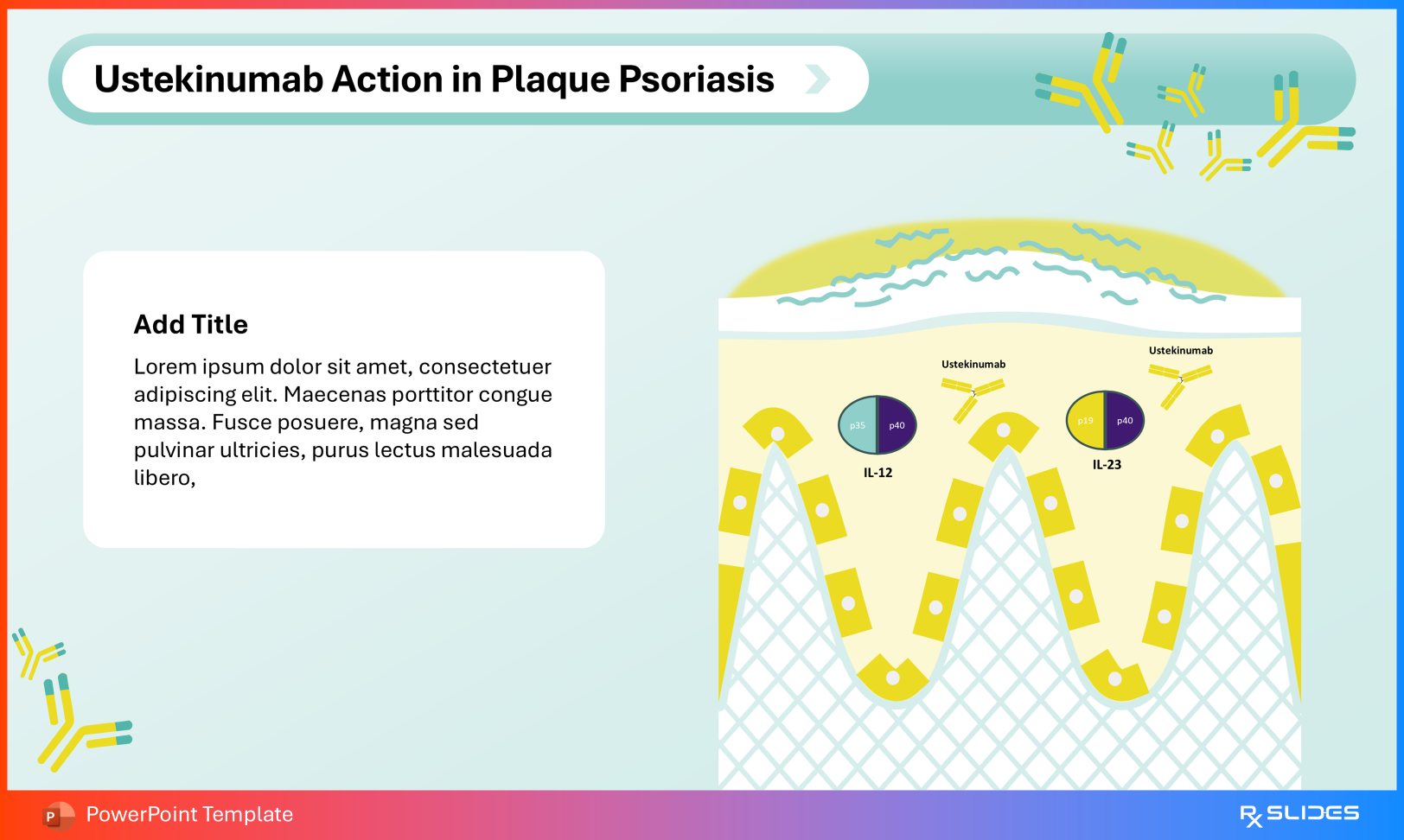
Plaque psoriasis Psoriatic arthritis Crohn’s disease Ulcerative colitis Indications Slide from Ustekinumab PowerPoint Presentation Template Disease-Specific Action Ustekinumab in Plaque Psoriasis Slide from Ustekinumab PowerPoint Presentation Template Dedicated visuals such as Ustekinumab Action in Plaque Psoriasis .
Ustekinumab in Crohn's Disease Slide from Ustekinumab PowerPoint Presentation Template Ustekinumab Action in Crohn’s Disease link cytokine inhibition directly to keratinocyte hyperproliferation or intestinal mucosal inflammation.
4. Administration and Dosing Strategies in Clinical Practice For infusion centers and nursing teams, operational clarity is critical.
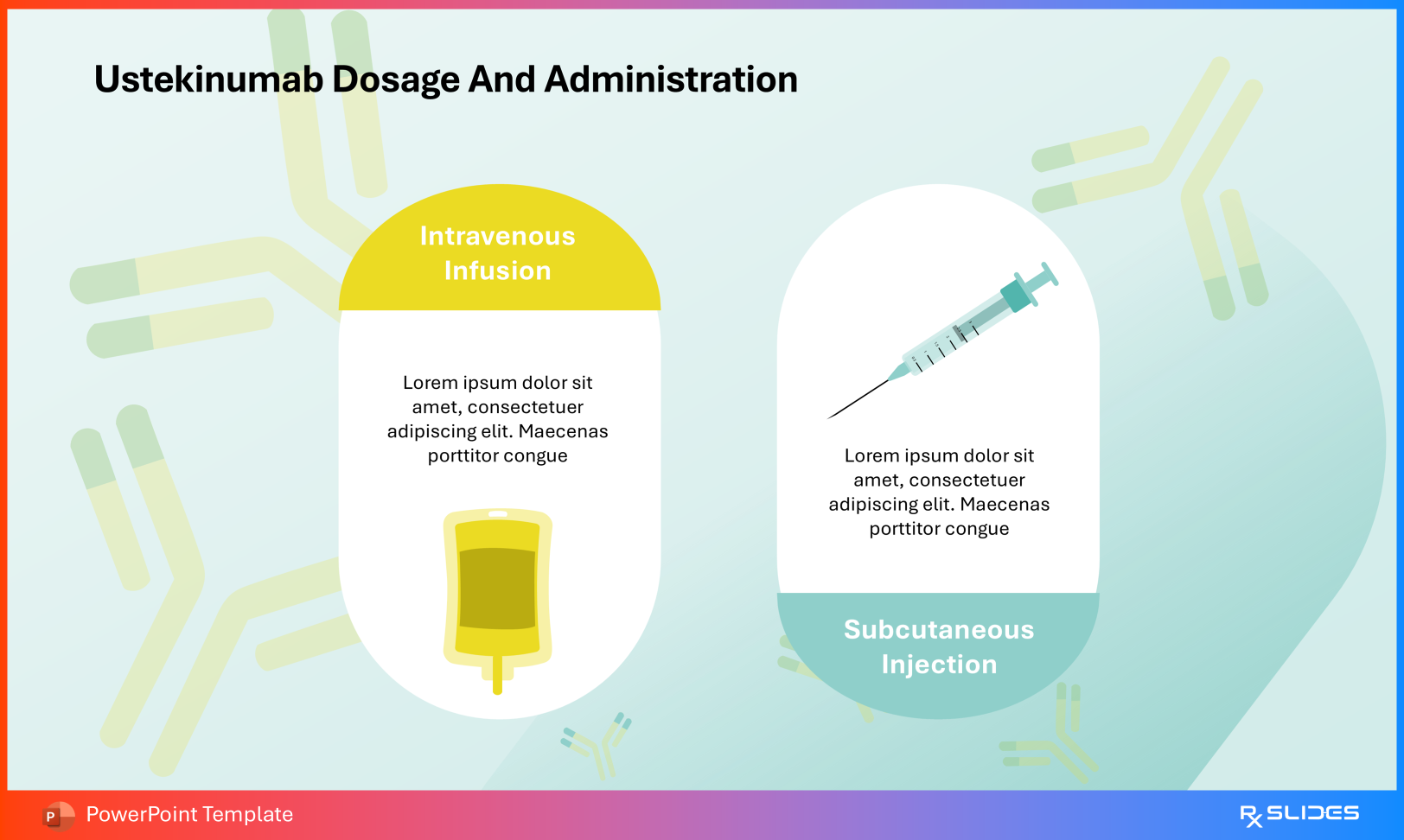
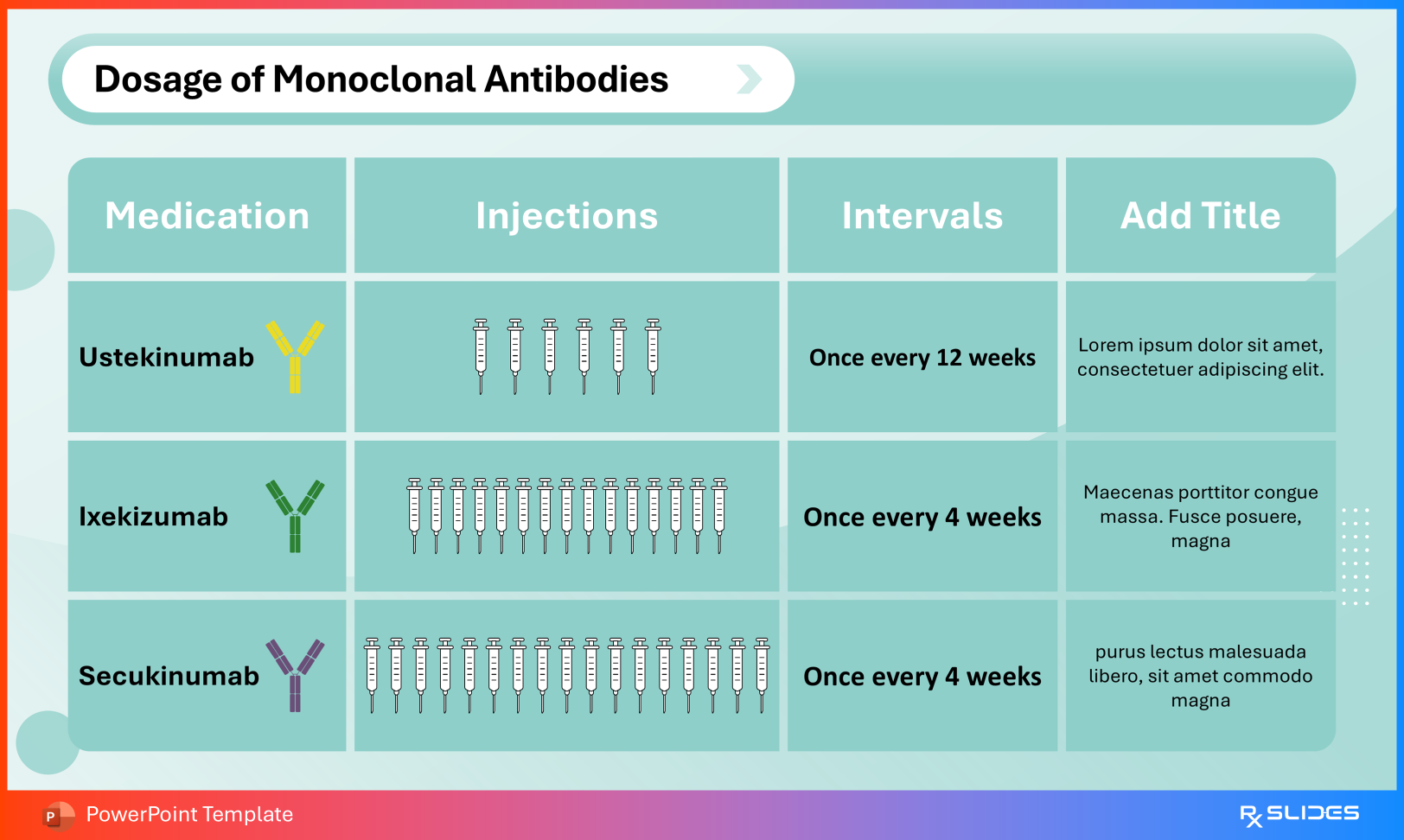
Dosage and Administration Use these slides to distinguish:
Intravenous infusion for induction (IBD indications) Subcutaneous injection for maintenance therapy Administration Slide from Ustekinumab PowerPoint Presentation Template Medication Intervals Visual timelines demonstrate dosing frequencies such as every 12 weeks versus every 4 weeks, supporting protocol adherence and patient scheduling.
Dosage Slide from Ustekinumab PowerPoint Presentation Template 5. Safety Profile: Warnings and Adverse Events Risk communication remains an essential component of biologic therapy education.
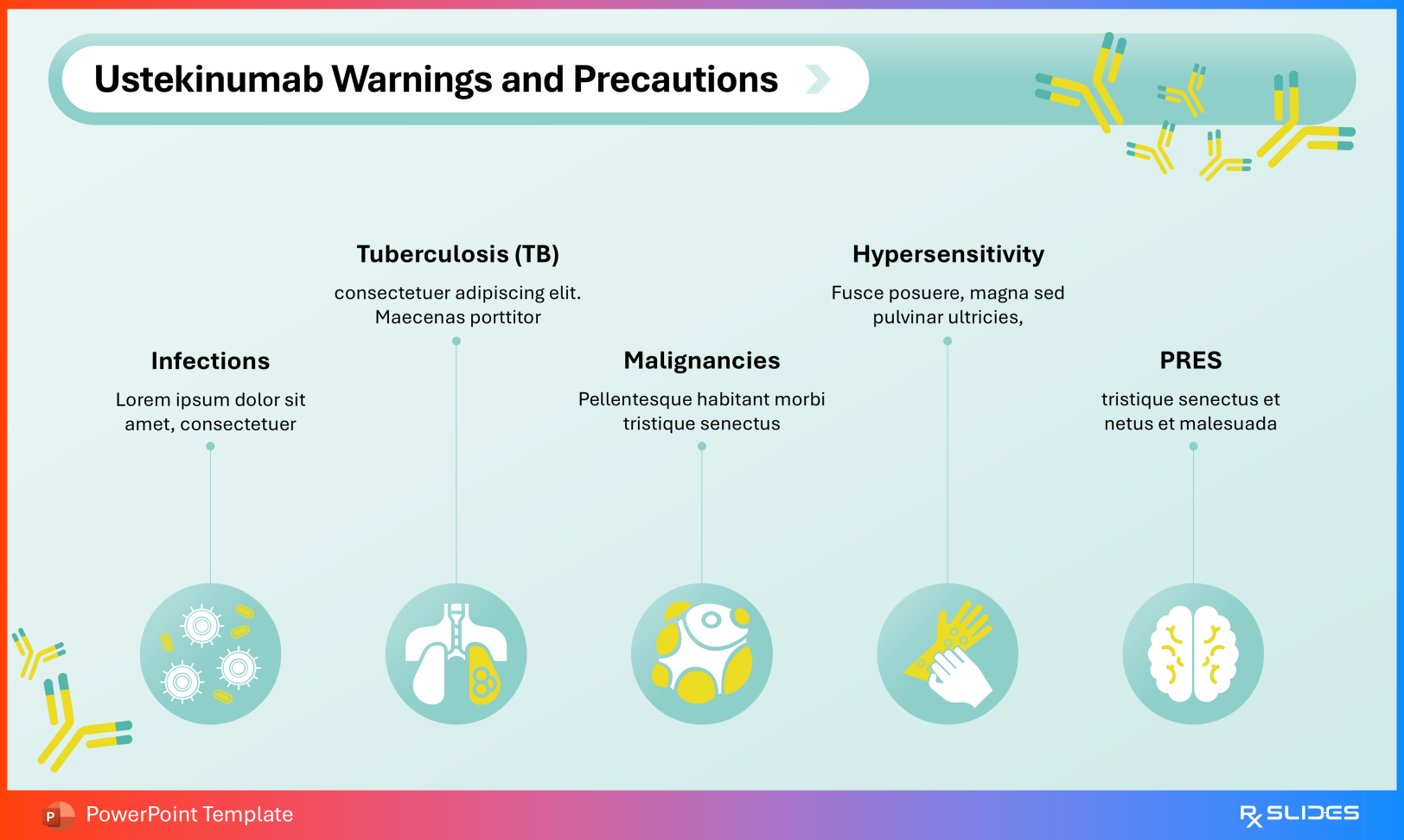
Warnings and Precautions Highlight serious considerations, including:
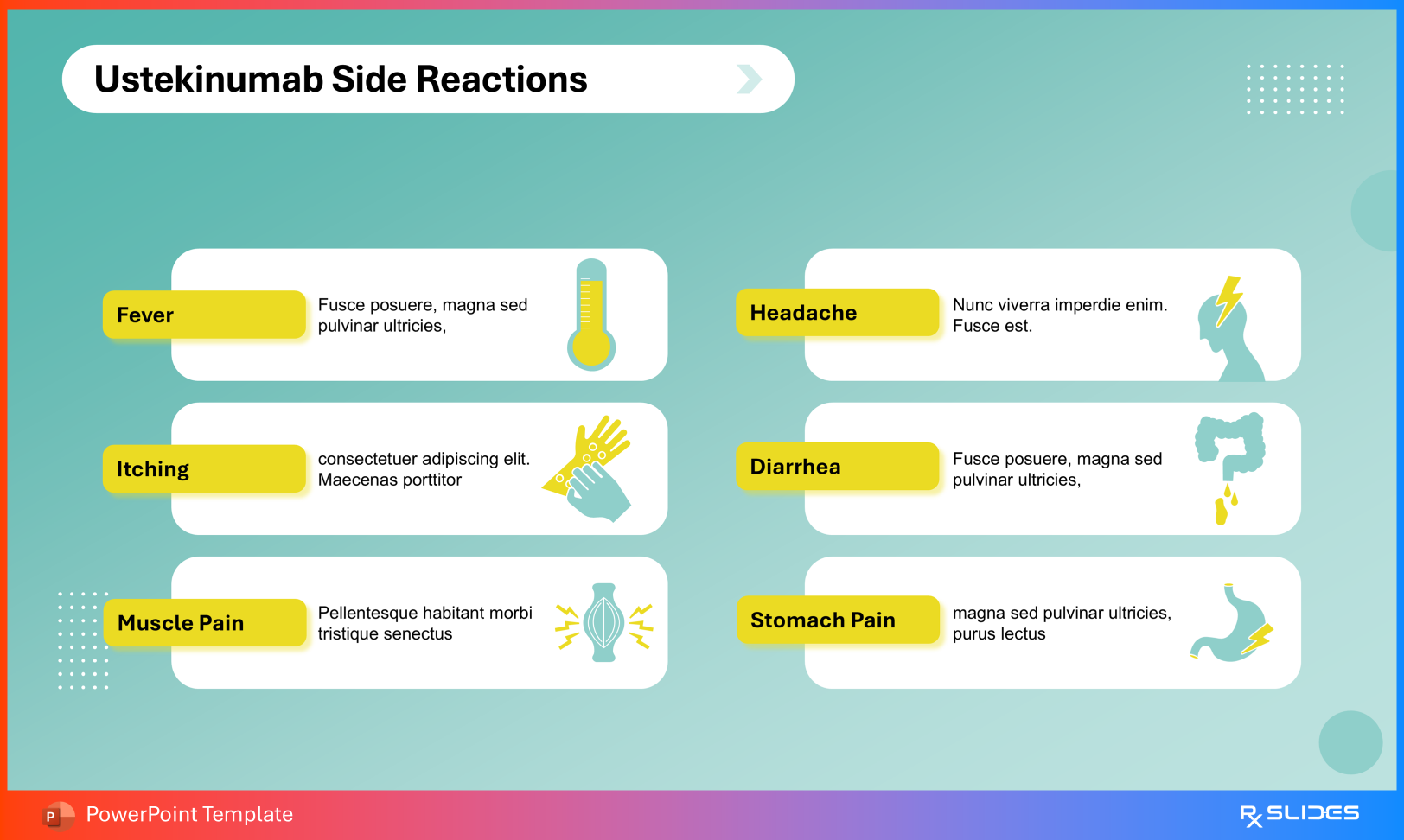
Tuberculosis reactivation Malignancy risk Hypersensitivity reactions Posterior Reversible Encephalopathy Syndrome (PRES) Precautions Slide from Ustekinumab PowerPoint Presentation Template Adverse Reactions Commonly reported effects include:
Headache Diarrhea Abdominal pain Fever Myalgia Side Reaction Slide from Ustekinumab PowerPoint Presentation Template These slides support informed prescribing and monitoring strategies.
6. Pharmacokinetics and Comparative Clinical Evidence For academic audiences and formulary committees, quantitative data drives decision-making.
Pharmacokinetics Use the Ustekinumab Distribution slide to illustrate:
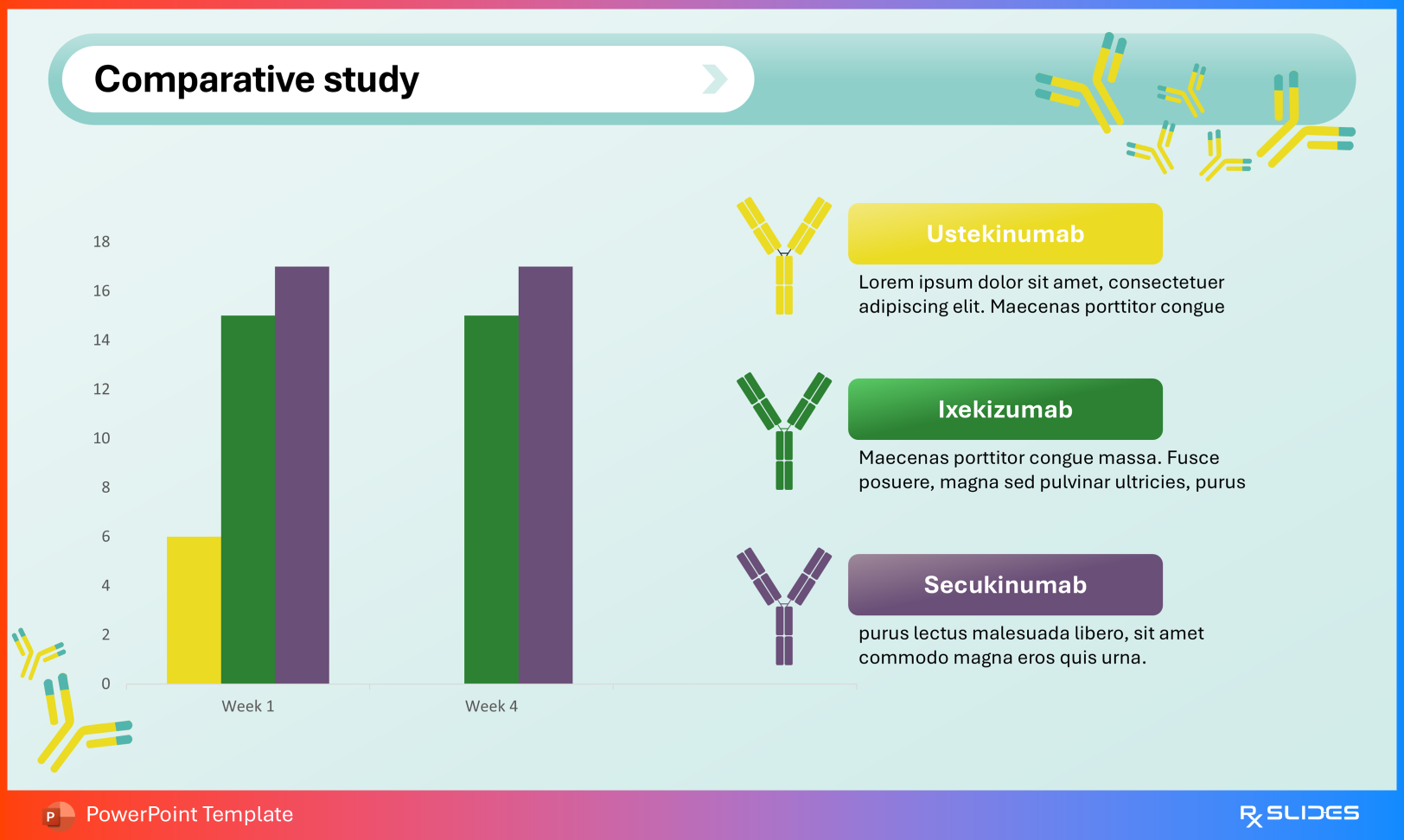
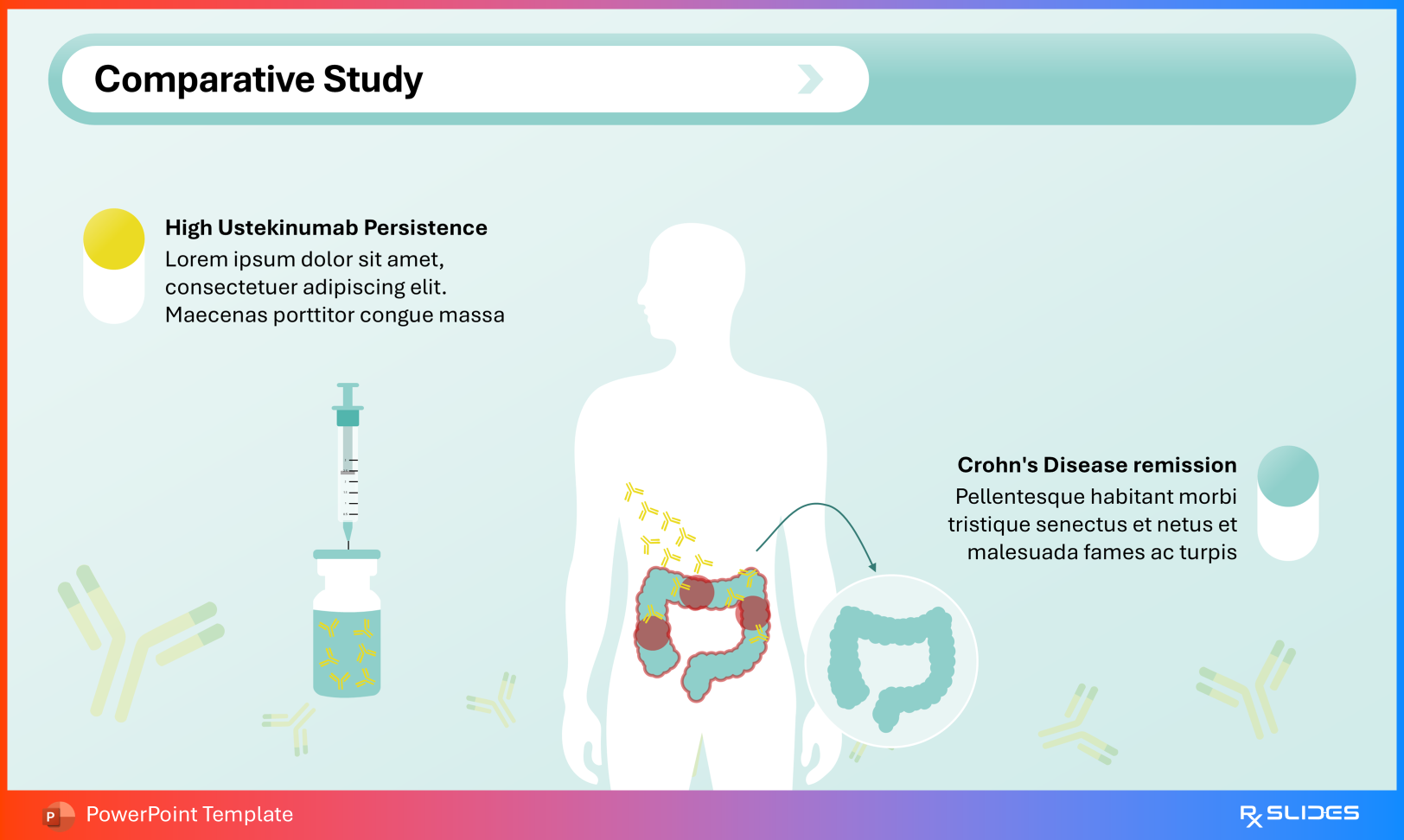
Volume of distribution: ~4.6 L in Crohn’s disease ~4.4 L in ulcerative colitis Terminal half-life approximately 3 weeks Distribution Slide from Ustekinumab PowerPoint Presentation Template Comparative Studies Benchmark ustekinumab against agents such as ixekizumab and secukinumab using the comparative efficacy slides.
Slide from Ustekinumab PowerPoint Presentation Template Clinical Outcomes Present remission and response data, including Crohn’s disease outcomes, to contextualize real-world effectiveness.
Remission and Response data Slide from Ustekinumab PowerPoint Presentation Template Conclusion: From Cytokine Subunits to Clinical Remission This template bridges the molecular geometry of the IL-12/23 p40 subunit with practical realities such as infusion protocols and remission rates. It supports seamless transitions from antibody variable regions to inflammatory bowel disease outcomes.
For healthcare professionals, it offers a coherent framework to teach biologic pharmacology, immune modulation, and evidence-based application across dermatology, rheumatology, and gastroenterology.